Would you like to make this site your homepage? It's fast and easy...
Yes, Please make this my home page!
HOME~~~PREV~~~NEXT
The following outlines explain the MECHANISMS of
growth PROMOTION by REDUCTANTS and
growth INHIBITION by OXIDANTS.
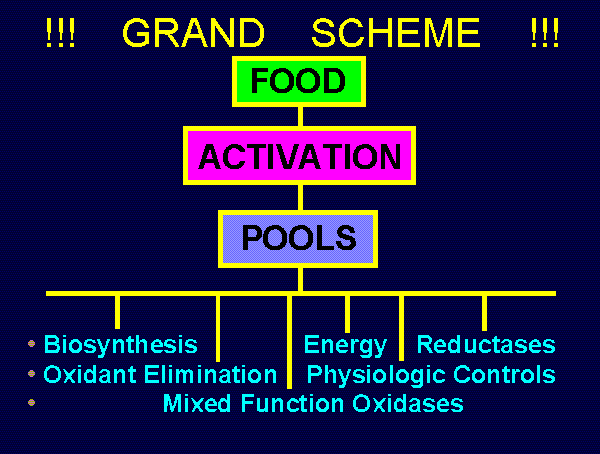
~~~ GRAND SCHEME OF MANAGING
THE DISTRIBUTION OF REDUCING EQUIVALENTS ~~~
FOOD SOURCES:
Carbohydrates, Fats, Proteins, Supplements
ACTIVATION MECHANISMS:
Glycolysis, Hexose Monophosphate Shunt,
Tricarboxylic Acid Cycle, Oxidative Detoxification
CARRIER POOLS:
NADH, NADPH, GSH, Certain Phenols
UTILIZATION CHANNELS:
myriad BETS
FUNCTIONS:
Synthesis, Energy Production, Reduction of Oxidants,
Various Reductases, Physiologic Controls,
Mixed Function Oxidases
Proliferation of cells requires a high level
of synthetic activity.
Many synthetic processes require ample supplies
of reducing equivalents.
Alteration of the relative demand of each
reductant consuming pathway will alter
the final distribution of reducing equivalents.
Diversion of reducing equivalents to energy production
or to oxidant removal depletes the carrier pool.
Depleted carrier pools result in decreased supplies
for synthesis.
Inhibition of synthesis inhibits proliferation.
~~~ KEY PROCESSES NECESSARY TO PROLIFERATION
WHICH DEPEND ON REDUCTANTS ~~~
activation of FOLATE to tetrahydrofolate:
needed to produce thymine from uracil etc.
activation of THIOREDOXIN:
needed to produce deoxyribonucleosides
activation of ORNITHINE DECARBOXYLASE:
needed to produce putrescine and polyamines
activation of GLYOXALASE by reduced glutathione:
needed to remove alpha-keto-aldehydes
activation of beta-ketoacyl-ACP-REDUCTASE,
crotonyl-ACP-REDUCTASE, other reductases:
needed to synthesize fatty acids
activation of beta-hydroxy-beta-methylglutaryl-
CoA-REDUCTASE:
needed to synthesize turpenoids and cholesterol
~~~ DISEASES ASSOCIATED WITH ~~~
PROLIFERATION & SYNTHESIS & REDUCTANT DEPENDENCY
= ALLERGIES: humoral
cellular
= INFECTIONS: viral
inclusion
bacterial
fungal
parasitic
= NEOPLASMS: benign
malignant
~~~ "OXIDATION-REDUCTION POTENTIALS
IN BACTERIOLOGY AND BIOCHEMISTRY" ~~~
by LF HEWITT, 6th Ed, E. & S. LIVINGSTONE LTD., 1950
TOPICS: oxidation-reduction processes
electrode potentials and polarography
redox indicator methods
effects of pH
biologic electron/hydrogen carriers
metabolic reductant production systems
lag phase versus log growth phase
peroxide resistance
bactericidal dyes, mold products, quinones
redox potentials of different bacteria
redox potentials and virulence
~~~ REFERENCES BY ALBERT SZENT-GYORGYI, MD, PhD, NI ~~~
BIOELECTRONICS - A STUDY IN CELLULAR REGULATION,
DEFENSE, AND CANCER 1968, Academic Press
THE LIVING STATE - WITH OBSERVATIONS ON CANCER
1972, Academic Press
THE LIVING STATE AND CANCER 1978, Marcel Dekker
TOPICS DISCUSSED:
- semiconductor characteristics of hydrated proteins
- pools of exchangeable hydrogen carriers
- how primitive and dividing cells require
an abundance of reductants
- phenol oxidases and peroxidases as defense systems
against infection in higher plants
- physiologic effects of ratios of alphaketoaldehydes
to thiols and to amines
- methylglyoxal as growth regulator
- mechanism of glyoxalase activation by reduced glutathione
BIOCHEMICAL PECULIARITIES OF TUMORS
WHICH CAUSE INTERNAL REDOSIS:
1) glucose / insulin receptors overabundant
2) hexose monophosphate shunt upregulated
3) glycerol-3-phosphate dehydrogenase deficient
4) cysteine sequestration upregulated
5) glutathione peroxidase deficient
BIOCHEMICAL ADVANTAGES OF REDOSIS
WHICH FAVOR TUMOR GROWTH AND SURVIVAL:
1) ensure sufficient hydrogen donors for synthesis
2) toggle transcription factors and growth factors
towards mode
3) enhance antioxidant protection against environment
4) enhance resistance to oxyradical attack by killer cells
5) maintain apoptosis triggers in mode
~~~ HOW TO DIVERT REDUCING EQUIVALENTS
TOWARDS ENERGY PRODUCTION ~~~
EXERCISE - - - - - ADP activates mitochondrial ETC function
increases perfusion
RIBOFLAVIN - - - - precursor to FAD oxidoreductases
THYROID & DHEA - - both induce G3PDH
QUINONES - - - - - shuttle reductants to cytochromes
HYDROGEN PEROXIDE - activates cytochromes
OXYGEN - - - - - - activates cytochrome A
COPPER - - - - - - ensures adequate cytochrome A
LIGAND DETOX * - - ensures active cytochrome A
AMMONIA DETOX - - - ensures active carbonyl carriers
MANGANESE - - - - - ensures adequate mitochondrial SOD
* HS- CN- NN- CO
~~~ HOW TO DIVERT REDUCING EQUIVALENTS
TOWARDS OXIDANT QUENCHING PATHWAYS ~~~
OXYRADICALS (bad choice):
radiation, selenium deficiency, toxic metals,
iron overload or quinone overdose
HYDROGEN PEROXIDE:
IV-H2O2, PO-H2O2, PO-MgO2
ozone, quinones, autoregeneration
LIPID PEROXIDES:
auto-oxidation of PUFA's,
ene reaction of singlet oxygen
OXIDES OF CHLORINE: ClO2 HClO HClO2 HClO3 HClO4
ULTRAVIOLET BLOOD IRRADIATION:
photoreduction consumes reductants
singlet oxygen produces peroxides and quinones
produces vitamin D a redifferentiator
COPPER: activates many endogenous oxidases
SELENIUM: activates glutathione peroxidase pathway
GLUTATHIONE: activates glutathione peroxidase pathway
activates reductive antioxidant family
activates oxidant sensitive immunoreceptors
~~~ HOW EXCESS ACCUMULATION OF REDUCTANTS OCCURS ~~~
CAUSES: MECHANISMS:
overeating - - - - - - - - substrates for NADPH production
inactivity - - - - - - - - depressed reductant consumption
hypothyroidism - - - - - - depressed reductant consumption
depressed GPDH shuttling
DHEA deficiency - - - - - elevates G6PDH activity
cortisol excess - - - - - elevated gluconeogenesis
insulin insensitivity - - elevated glucose
Se deficiency - - - - - - failure of glutathione peroxidase
failure of T4 to T3 conversion
Cu deficiency - - - - - - failure of numerous oxidases
carbon monoxide - - - - - blocks Hb and CytA
hydrogen sulfide - - - - - blocks CytA and other Cu enzymes
xenobiotic exposure - - - depletes glutathione
induces quinone reductases
~~~ HOW TO STARVE THE SUPPLY SIDE ~~~
LOW CALORIE DIET
FASTING
EXERCISE
CORRECT HYPERGLYCEMIA
HYDRAZINE SULFATE
GLUCOCORTICOID AVOIDANCE
G6PDH DEFICIENCY
DHEA
SILVER
XENOBIOTIC AVOIDANCE
MAGADOSE REDUCTANT AVOIDANCE
HOME~~~PREV~~~NEXT
